|
1/29/2024 0 Comments Aami tir452012 r 2018 pdf 
It gives a list of recommendations throughout the documents. The whole document is in fact a discussion on how aligning the regulatory perspective and the agile perspective. However, both can be aligned - or reconciled - at the level of perspectives (or goals), concepts, and practices. This is what generates a lot of concern about agile methods in a regulated environment. The one thing to remember of this TIR, is that the regulatory perspective and the agile perspective are not aligned at first sight. To achieve this goal, a verb is continuously repeated in the document: to align. YES, they allow to reach the level of scrutiny demanded by regulatory framework.YES, agile methods can be used to develop medical device software,.Given the concerns of many manufacturers about agile methods, this technical report is here at first to reassure them. 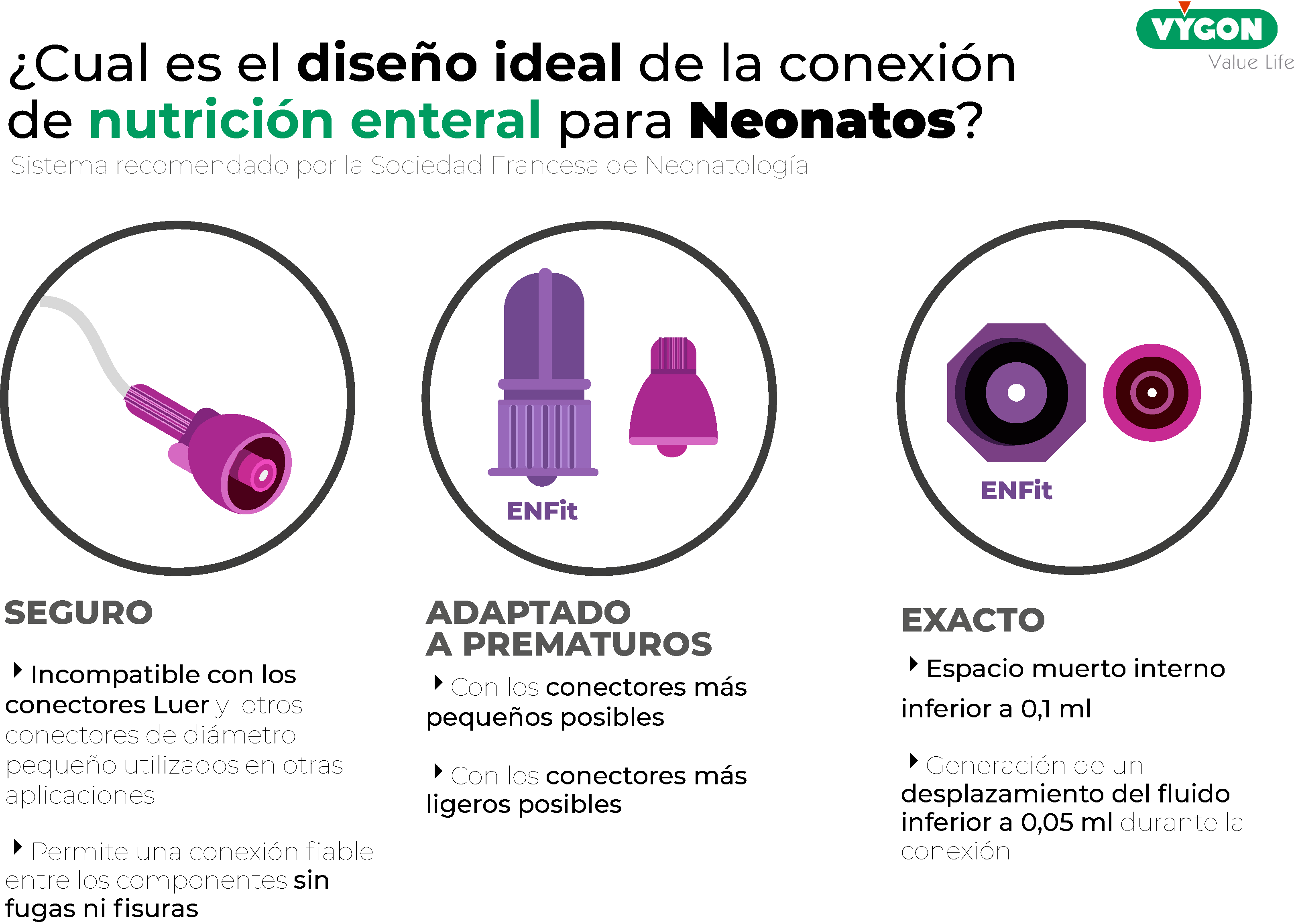 
I read this technical report of the AAMI (yes, I did! No kidding!). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
